How Genetic Testing Changes Your Treatment
The core goal of this technology is to maximize a drug's benefits while minimizing Adverse Drug Reactions (ADRs). These reactions aren't just mild side effects; they are serious enough to cause about 6.7% of all hospital admissions. When a doctor orders a pharmacogenomic (PGx) test, they are looking for specific variants in your DNA that tell them how you metabolize a drug.
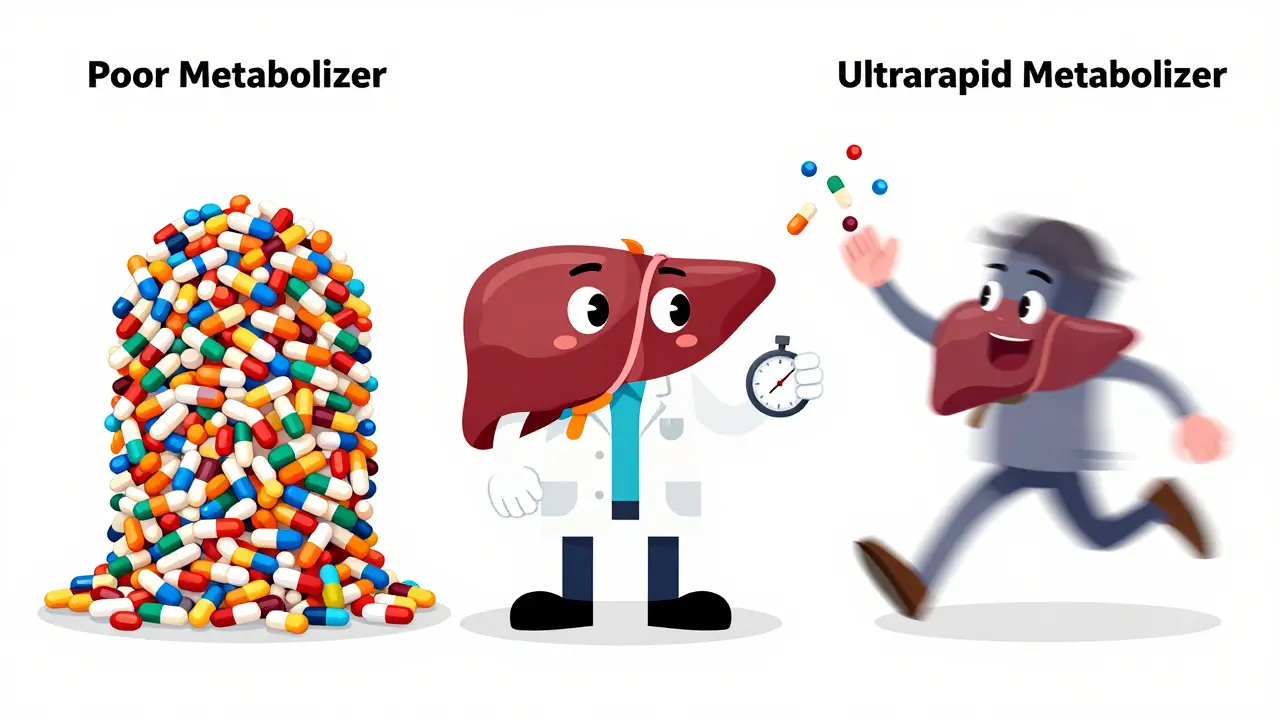
Most of these tests focus on the Cytochrome P450 system, a family of enzymes in the liver. If you have a genetic variant that makes you a "poor metabolizer," a drug might build up in your system, leading to toxicity. On the flip side, an "ultrarapid metabolizer" breaks down the drug so quickly that it never reaches a therapeutic level in the blood, meaning the medicine simply doesn't work.
For example, consider the gene CYP2D6. This enzyme processes everything from codeine to certain antidepressants. A person who is a poor metabolizer might experience severe nausea and no pain relief from codeine, whereas an ultrarapid metabolizer could potentially experience an overdose from a standard dose because their body converts the drug into its active form too quickly.
The Practical Process: From Swab to Prescription
Getting a PGx test isn't as invasive as you might think. You don't need a surgical biopsy. Most clinical platforms, such as those used by the Mayo Clinic, collect DNA through a simple saliva sample, a blood draw, or a quick cheek swab. Once the sample is sent to the lab, technicians look at a panel of clinically relevant genes. While there are over 100 genes that could affect drug response, most tests focus on a core group-like CYP2D6, CYP2C19, and CYP2C9-that account for 70-80% of significant drug-gene interactions.
Once the results are in, doctors use guidelines from organizations like the Clinical Pharmacogenetics Implementation Consortium (CPIC). CPIC provides the evidence-based "rulebook" for how to change a dose based on a genetic result. For instance, if a patient carries the HLA-B*15:02 variant, the risk of a life-threatening skin reaction called Stevens-Johnson Syndrome when taking carbamazepine is nearly 1,000 times higher. In this case, the genetic test isn't just a suggestion-it's a critical safety barrier.
| Gene | Associated Medication | Impact of Genetic Variant | Clinical Action |
|---|---|---|---|
| CYP2C19 | Clopidogrel | Poor metabolizers cannot activate the drug | Use alternative antiplatelet therapy |
| CYP2D6 | Paroxetine / Codeine | Varying speeds of metabolism (Poor to Ultrarapid) | Adjust dose or switch medication |
| HLA-B*57:01 | Abacavir | High risk of severe hypersensitivity reaction | Avoid drug entirely |
| SLCO1B1 | Simvastatin | Increased risk of muscle damage (myopathy) | Lower dose or use different statin |
Where This Works Best (and Where It Doesn't)
Pharmacogenomics isn't a magic bullet for every pill in your cabinet. Its effectiveness varies wildly depending on the medical specialty. In oncology, it's already a standard; about 62% of institutions use it to match patients with targeted therapies that attack specific mutations in a tumor. Psychiatry is another huge win. Data shows that patients using PGx-guided treatment for major depressive disorder have a remission rate of 30.8%, compared to just 18.5% for those using standard trial-and-error methods.
However, there are gaps. In cardiology, the results are more mixed. The TAILOR-PCI trial showed that using CYP2C19 testing for clopidogrel didn't significantly reduce cardiovascular events compared to standard therapy. This tells us that while genetics are a huge piece of the puzzle, they aren't the only factor. Your age, weight, kidney function, and other medications you're taking also play a role.
There's also the issue of "actionability." Currently, only about 15-20% of commonly prescribed medications have clear, evidence-based genetic guidelines. If you test for a gene that doesn't have a corresponding CPIC guideline, your doctor might tell you the result is "informative but not actionable," meaning it doesn't actually change your treatment plan.

Overcoming the Hurdles to Adoption
If this tech is so great, why isn't every doctor doing it? The biggest barrier is infrastructure. Most healthcare systems struggle to integrate genetic data into electronic health records (EHR). Imagine a doctor prescribing a drug today, but the genetic warning is buried in a PDF from a test taken three years ago. For a system to work, the EHR needs to trigger an automatic alert-a "clinical decision support" pop-up-the moment a drug is typed into the system.
Cost and insurance are the other big hurdles. While a test might cost under $25 for some specific markers in a research setting, comprehensive panels are more expensive. Insurance coverage is uneven; you're much more likely to get a PGx test covered if you have cancer than if you're treating depression. This creates a gap where only those who can afford out-of-pocket testing get the benefit of personalized dosing.
Finally, there is a diversity problem. About 78% of pharmacogenomics studies have been conducted on people of European ancestry. This means the "standard" guidelines might not be as accurate for people of African, Asian, or Hispanic descent. Programs like the NIH All of Us Research Program are trying to fix this by collecting data from millions of diverse participants to ensure personalized medicine is actually personal for everyone.
Looking Ahead: The Future of Your Medicine Cabinet
We are moving toward a world of "preemptive genotyping." Instead of testing you after you fail a medication, doctors will test you once, early in life, and store your genetic profile in your medical record. Whenever you need a new prescription-whether it's for high blood pressure, anxiety, or pain-the system will automatically check your DNA and suggest the safest option.
The FDA is already pushing this forward, with draft guidance proposing mandatory testing for several more drugs, including certain statins and SSRIs, by 2025. We are also seeing the rise of polygenic risk scoring, which looks at multiple genes instead of just one to predict drug response more accurately. By 2027, experts predict that half of all commonly prescribed medications will have actionable genetic information.
Is pharmacogenomic testing the same as a 23andMe test?
Not exactly. While direct-to-consumer tests can identify some variants, clinical PGx tests are usually more comprehensive and use higher-grade analytical sensitivity (often 99%+) to ensure accuracy. Clinical tests are interpreted by medical professionals using CPIC guidelines, whereas consumer tests provide general information that may not be actionable for a prescribing doctor.
Will this test tell me if a drug will work for me?
It tells you about your capacity to process the drug. It can tell you if you are likely to have a severe side effect or if the drug will be ineffective because you metabolize it too quickly. However, it cannot guarantee a drug will work, as other factors like your lifestyle, diet, and other health conditions also influence your response.
Does the test hurt?
No. Most PGx tests are non-invasive. They typically require a cheek swab (rubbing a cotton swab inside your mouth), a saliva sample, or a standard blood draw. None of these procedures are painful.
How long do the results stay valid?
Your DNA doesn't change. Once you've had a pharmacogenomic test, the results are generally valid for your entire life. What does change is the science-as new research emerges, a variant that was "unknown" five years ago might become "actionable" today.
Can I request this test from my doctor?
Yes. If you've had a history of bad reactions to medications or if you're starting a drug known to have high genetic variability (like certain antidepressants or blood thinners), ask your provider about PGx testing. Be prepared to check with your insurance company regarding coverage, as it varies by medication type.



